Colorectal Adenomatous Polyp (CAP) was one precursor of colorectal most cancers (CRC) and having a excessive likelihood of creating into CRC. There was a lack of conclusive chemoprevention evidences to prevention new CAP occurrence in post-polypectomy.
Xiaoai Jiedu Decoction, Chinese National Medical Professor (Zhou Zhongying)’s expertise components, has been used to deal with new CAP occurrence in post-polypectomy from the 20th century in China.
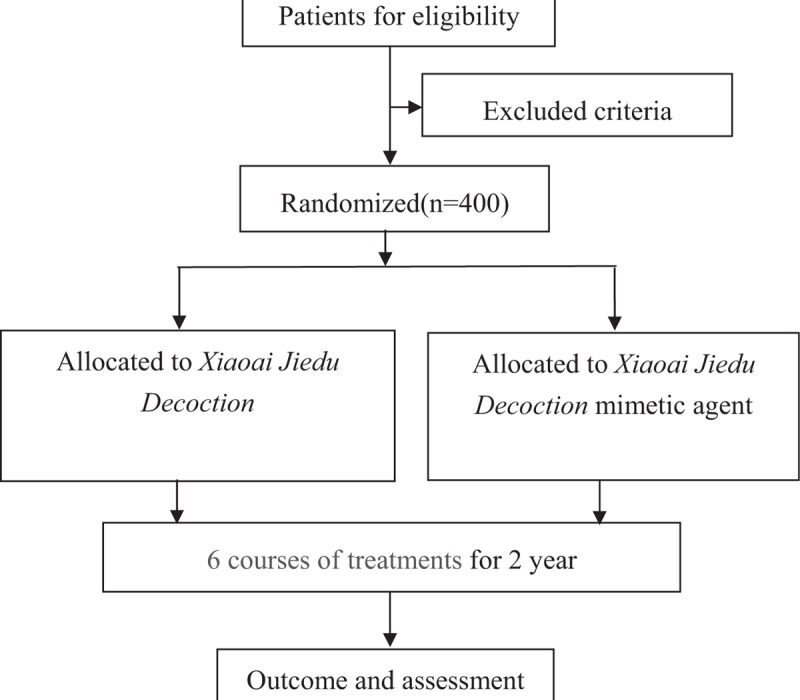
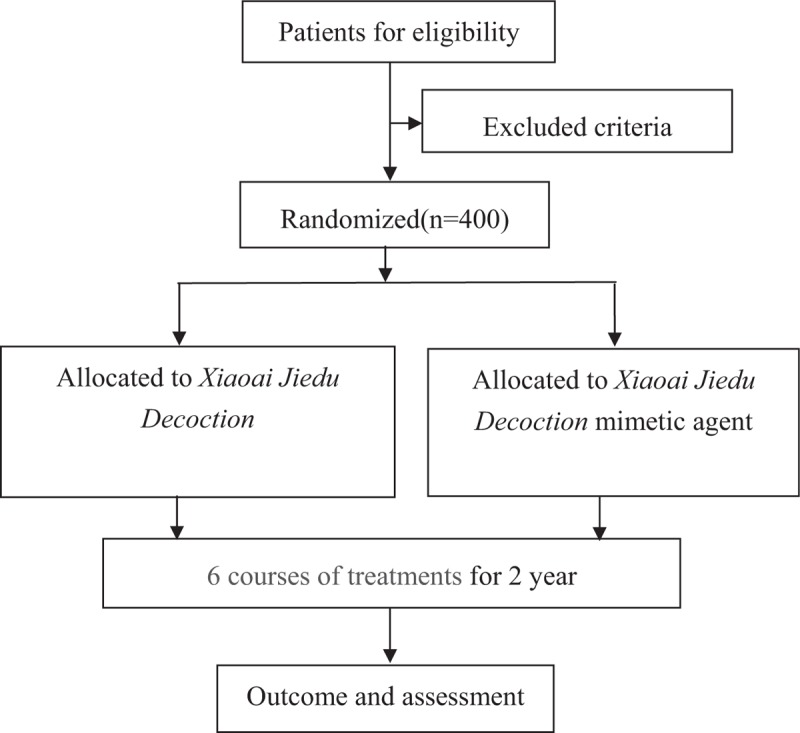
However, medical analysis of Xiaoai Jiedu Decoction in the treatment of CAP recurrence was lack. We design this research to judge the efficacy and security of Xiaoai Jiedu Decoction in the treatment of new CAP occurrence in post-polypectomy on colonoscopy.A randomized, controlled, blind and multicenter trial to judge the efficacy and security of Xiaoai Jiedu Decoction is proposed.
CAP sufferers (after full polypectomy underneath colonoscopy) will likely be randomly assigned into Xiaoai Jiedu Decoction group and Xiaoai Jiedu Decoction mimetic agent group. Patients will obtain 6-course remedies and a 2-year follow-up.
Follow-up colonoscopy will likely be anticipated to carry out in 1 and a couple of years after the baseline examinations. The major final result measure is the new CAP occurrence in 1 and a couple of years.
The secondary final result measure is the occurrence of superior adenoma in 1 and a couple of years.This research will present goal evidences to judge the efficacy and security of Xiaoai Jiedu Decoction as an adjuvant treatment for new CAP occurrence in post-polypectomy.NCT03616444.
Circulating Myeloid Derived Suppressor Cells (MDSC) That Accumulate in Premalignancy Share Phenotypic and Functional Characteristics With MDSC in Cancer.
Myeloid derived suppressor cells (MDSC) are a heterogeneous inhabitants of immature myeloid cells that accumulate in circulation of most cancers sufferers and at tumor websites the place they suppress anti-tumor immunity.
We beforehand reported that in a colon most cancers preventiontrial of a MUC1 vaccine examined in people at elevated danger for colon most cancers, those that didn’t mount immune response to the vaccine had larger pre-vaccination ranges of circulating MDSC in comparison with those that did.
We additionally reported that people with pancreatic premalignancy, Intraductal Papillary Mucinous Neoplasm (IPMN), had elevated circulating ranges of MDSC that inversely correlated with spontaneous antibody responses towards the pancreatic tumor related antigen MUC1, abnormally expressed on IPMN.
Accumulation of MDSC in most cancers and their immunosuppressive position had been properly established however their presence in premalignancy was sudden. In this research we in contrast MDSC in premalignancy with these in most cancers with the speculation that there is perhaps variations in the composition of varied MDSC subpopulations and their immunosuppressive features resulting from totally different lengths of publicity to illness and/or totally different tissue microenvironments.
In cohorts of sufferers with premalignant polyps, colon most cancers, premalignant IPMN, and pancreatic most cancers, we confirmed larger ranges of MDSC in premalignancy in comparison with wholesome controls, larger ranges of MDSC in most cancers in comparison with premalignancy, however no distinction in their subpopulation composition or immunosuppressive capability.
We present that ranges of MDSC in premalignancy correlate negatively in vivo with spontaneous MUC1-specific antibody responses and in vitro with polyclonal T cell proliferation and IFN-γ secretion.